❶ Give us your email address and you'll be sent an Aggressive Risk Management Method PDF for FREE. It combines FMEA and Fault Tree Analysis (FTA) to iteratively and incrementally lower failure mode probabilities. That will find and control more faults.
❷ Interview and you'll get help implementing the method for FREE (plus expenses). It will include a PowerPoint presentation using the Method PDF.
❸ In addition, you get a peek into the future at automatic FDA regulatory compliance.
❸ In addition, you get a peek into the future at automatic FDA regulatory compliance.
❹ The interview also makes you a Menlo Park Associate, entitled to VOTE on new and deferred requirements. Don't miss getting your needs addressed in the new software. Then ➄ come to the Associates cast party.
4. Use the software for free and FOREVER by interviewing and seeing the DEMO so as to hasten development and secure funding.
3. Use the software for FREE as long as it is needed for the pandemic.
2. Improve risk control and prevent device failures and recalls while observing social distances and practices.
1. Stop manual / outsourcing of compliance, and apply that effort towards production of devices for critical pandemic health care.
Be the hero of your own story and save your enterprise!
FDA Recall 'Epidemic'?
|
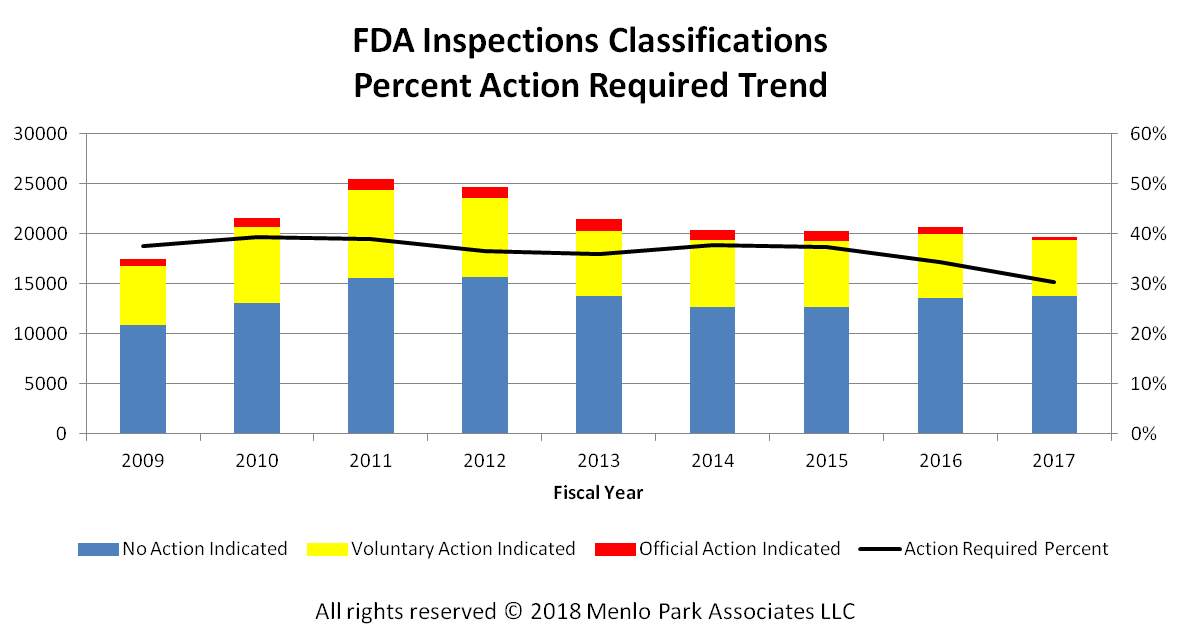
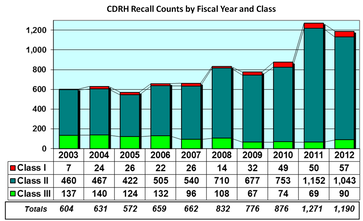
The chart at right started the 'problem'. An FDA report (Medical Device Recall Report FY2003 to FY2012) had this chart and later the comment "The annual number of medical device recalls increased by 97 percent from FY 2003 to FY 2012".
Fast forward to 2016 and a Medical Device and Diagnostic Industry magazine cover article, titled "Medtech’s Recall Epidemic" reports a record high number of FDA medical device recalls in 2014. The authors, Joshua R. Dix, Suraj Ramachandran, and Darin S. Oppenheimer, blame medical device manufacturers and their poor risk management.
|
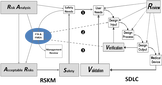
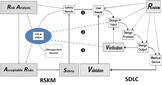
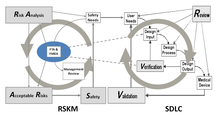
The real crisis is that new device company risk management is not any better than what older companies do. They all struggle with an old classic practice. The industry needs to find and learn a new, natural and easier way to do risk management that is more aggressive and probes deeper. The videos have the secret.
I’m a management consultant researching the impact of the current FDA 'recall epidemic' on individual firms. 21st century social media software technology can lower the number of recalls, lower the effort and costs to avoid them and give confidence they are prevented, and it’s not just good luck they don't happen.
|
'Epidemic' vs. Economics
|
The FDA Medical Device Recalls chart above shows what's really going on. There has been a rise in recalls since 2012, but this correlates 95% with the number of new medical device introductions. More devices means more recalls. It's more economic than epidemic.
|
My solution has a new method to lower your risks and save lives. It combines FMEA and Fault Tree Analysis to drive down medical device failure probabilities and can be plugged into your existing processes and practices. I have software that turbocharges your risk management and compliance.
|
FDA Recall Prescription
• Integrated project and risk teams that easily create risk management synergy as a natural byproduct of their work.
• Aggressive risk management using Detection that naturally finds and mitigates more faults and drives down failure probabilities.
• Iterative approach to easily mitigate known faults and incrementally find unknown failure modes to exceed global and proprietary standards of quality and safety.
• Embedded learning program to easily leverage knowledge of past risks and naturally contribute new risks and information as corporate proprietary assets.
• Internal tracking of product fault/failure modes and project risks for natural meetings, for reporting and for interfaces required by analysis.
• Interfaces to software via API for Failure Mode and Effects Analysis (FMEA) and Fault Tree Analysis (FTA).
|
FDA Compliance PrescriptionA 21st Century project management platform using social media software technology that holds and enforces an organization's Quality Management System, supports medical device manufacture using aggressive and iterative risk management, and that generates project communications and compliance documentation for history files and records.
• Digital and customizable procedures
• Easy task tracking to completion • Procedure tracking to completion • Internal chat channels create a permanent record that documents compliance in the logs of each issue, task and project, etc. that eliminate all email dumps for audits. • Automatic stakeholder alerts for reviews, etc.
• Automatic due reminders and update alerts that can also become external emails and text messages. • Integrated document management for each issue, task, meeting, report, project, and community or QMS. • Outsourcing will stop being the means of enforcing compliance.
|
Organization Prescription• Menlo Park Project Accounts sponsor 'Communities'. Communities own projects, which own Issues and Tasks. Communities select their default language and options for new projects. Their Documents Store holds controlling documents at the top of the configuration management tree.
• Menlo Park Project supports formal project management practices like generating detailed reports and meeting documents as well as ad hoc messaging.
|
Project Prescription• Ability to lock versions of controlling documents so projects finish in a stable environment, unimpeded by changes going on around them.
• Controlling documents in the Documents Store are shown by default for all Project objects like Issues and Tasks.
• Thus, project work proceeds normally and naturally without confusion as to plans, policies, or other guidance documents.
|
Startup Prescription
• Menlo Park Project Digital SOP's will replace out-sourcing as the means to establish procedures, policies, objectives, controls, authorizations, training and other resources including individual Part 820 subparts.
• Digital SOP's may be available for purchase for common types of devices. Or, already available written procedures may be translated into DSOP's and used many times over.
• Natural design, development and manufacturing activities using Menlo Park Project will document their own concordance with procedures and policies, etc. and corresponding regulatory compliance.
• Menlo Park Project will generate detailed reports and meeting agenda and minutes documents. Messaging available on most system objects become part of its history and show up in reports and meeting documents.
• Detailed reports, meeting documents and messages, plus the online meeting facility for remote participation allow outsourced resources to share their expertise and richly participate in project risk management and compliance activities.
• This keeps project costs low while maintaining maximum collaboration, synergy, and results. |
Project Manager Prescription
• Effective and efficient natural meetings.
• Comprehensive and efficient reporting. • Meeting design with automatic agenda and minutes generation. • Generated reports and meeting agenda and minutes automatically have copies saved in their individual Documents Store histories.
• Changes and comments automatically generate messages to interested team members and stakeholders who may choose to have them forwarded as emails or text messages.
PMO Prescription
• Ability to assign new [pilot] versions of control documents to test them in a sandbox environment and not bother other projects around them.
Team Member Prescription
• Issue and Task To-Do lists for prioritization, optimization, and collaboration.
• Relay of chat messages to team member or stakeholder email and texts via API Outsourcing Prescription
• Outsourcing will stop doing much of the manual work and chasing after people to complete compliance activities. It will be used for more consulting, based on particular expertise and proprietary assets, to tailor them for clients to guide development, set up SOP's and transition startup firms to production.
|
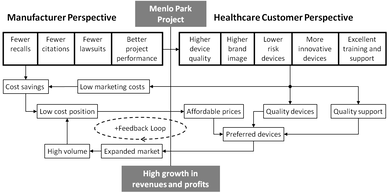
Better risk managementFewer device defects
Fewer FDA recalls Less anxiety
Cost savings Improved quality Better industry reputation |
Better compliance
More timely reviews
Fewer errors Fewer 483 observations Fewer warning letters No more Email dumps
Less anxiety Less rework Cost savings Improved quality |
Better performance
Less waiting (automatic alerts)
Better task performance More frugal outsourcing Fewer errors Less anxiety
Less rework Fewer missed deadlines Better estimating Cost savings Improved quality |
Recipients
|
Employees
Stakeholders C-level officers Board members |
Quality/RA Managers
Employees Stakeholders |
Project Managers
Project Manager Organizations Team members Stakeholders |
Rick Bollinger, Founder and CEO of Menlo Park Software
|
See the Founder's story of discovery in the video above. As a process improvement management consultant he was primed to recognize opportunities for innovation. The secret to better risk management is in the videos. Use the method he designed to harness it and the software he designed to support it and also help with compliance. Check below to get the details. And, maybe get a demo and give feedback.
Turn the Prescriptions and Benefits into reality with your demo and feedback.
|
Rick Bollinger has over 35 years of software development, project management, and consulting experience. Certified as a Project Management Professional and Six Sigma Green Belt, his broad experience includes many industries. He has consulted and done training to help companies like General Motors, Lockheed Martin, and Pfizer, and also government agencies like the Department of Defense in the U.S. and the Ministry of Health in Canada.
Rick has two degrees from the University of Michigan: a Bachelor's degree in industrial engineering (cum laude) and a Master's degree in computer engineering. And has been active in process improvement and speaks at various seminars and conferences of the PMI and the Dr. W. Edwards Deming Institute.
In 2012, after work and traveling, Rick found time to write a business plan (Winning $1,000 for his New Business Idea in the Phase I GLEQ [MiQuest] Business Plan Competition, Third Place). The company is named after Menlo Park, New Jersey, where Thomas Edison had his famous laboratory using the technology of his day to make life better and easier for people in his world.
|